Effect of Pressure on Aqueous Urea/Ammonium Nitrate Fuel Combustion
Bar Mosevitzky, Chemical Engineering, Technion, Haifa, Israel
Michael Epstein, Chemical Engineering, Technion, Haifa, Israel
Gennady E. Shter, Chemical Engineering, Technion, Haifa, Israel
Gideon S. Grader, Chemical Engineering, Technion, Haifa, Israel
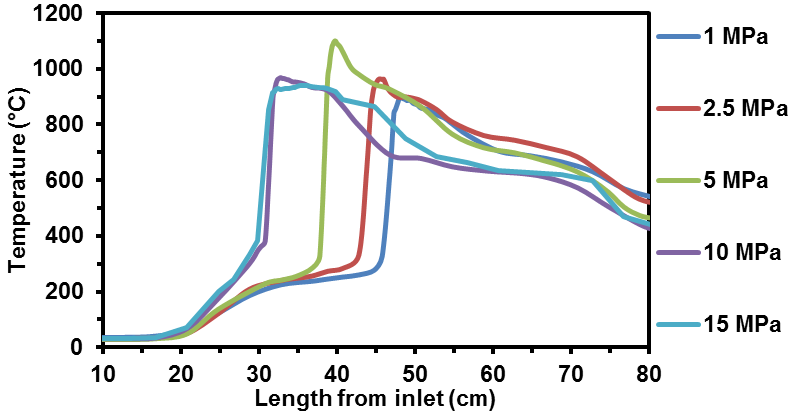
Fluctuations in renewable energy production inhibit its large-scale implementation. This intermittency can be overcome by energy storage. Chemical fuels exhibit high energy densities, making them ideal energy storage media. Specifically, aqueous solutions of urea and ammonium nitrate (UAN) can act as low-carbon synthetic fuels suitable for energy storage applications. Previous studies have demonstrated that UAN combustion generates CO, NOx and NH3. However, the mechanism involved in their formation was unclear due to complex reactor geometries and poorly characterized temperature profiles. In this work, a newly constructed tube reactor with a built in thermowell was used to study the effect of pressure on the continuous combustion of UAN and its resulting temperature profile (Fig. 1).
Figure 1. The effect of pressure on the inner UAN combustion temperature profile at 10 ml min-1 fuel flow rate. |
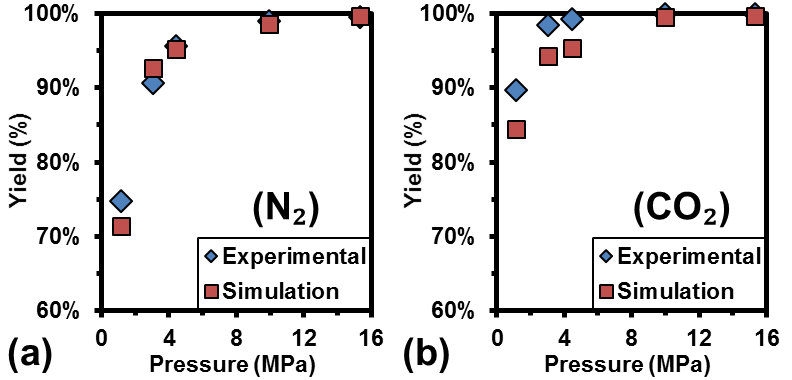
Experimental results indicated that lower pollutant emissions and higher product yields were achieved as pressure was increased from 1 MPa to 15 MPa (Fig. 2). An updated mechanism was applied to simulate the combustion of UAN at 1-15 MPa. Experimental and simulation nitrogen species concentrations were in excellent agreement at all investigated pressures. Carbon species levels were in excellent agreement at pressures above 5 MPa. These results were used as the basis for performing rate-of-production and sensitivity analyses. High pressures reduced the complexity of the reaction network, and lowered the sensitivity of the pollutant concentrations to the reaction kinetics, demonstrating the mechanism's robustness at high pressures. |
Figure 2. The effect of pressure on the yield of: (a) N2 and (b) CO2, as indicated by experimental (♦) and simulation (■) results. |
The effects of UAN combustion pressure on the reactor temperature profile and effluent composition will be presented, and the implications of the performed simulations and analyses on the reaction pathways of UAN combustion will be discussed.
Organized & Produced by:

POB 4043, Ness Ziona 70400, Israel
Tel.: +972-8-9313070, Fax: +972-8-9313071
Site: www.bioforum.co.il,
E-mail: bioforum@bioforum.co.il